Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ueki, Tadamasa; Niwa, Masakazu; Iwano, Hideki*; Danhara, Toru*; Hirata, Takafumi*
Chishitsugaku Zasshi, 125(3), p.227 - 236, 2019/03
no abstracts in English
Lee, C. G.; Iguchi, Kazunari; Inagawa, Jun; Suzuki, Daisuke; Esaka, Fumitaka; Magara, Masaaki; Sakurai, Satoshi; Watanabe, Kazuo; Usuda, Shigekazu
Journal of Radioanalytical and Nuclear Chemistry, 272(2), p.299 - 302, 2007/05
Times Cited Count:47 Percentile:93.88(Chemistry, Analytical)no abstracts in English
Usuda, Shigekazu; Yasuda, Kenichiro; Kokubu, Yoko; Esaka, Fumitaka; Lee, C. G.; Magara, Masaaki; Sakurai, Satoshi; Watanabe, Kazuo; Hirayama, Fumio; Fukuyama, Hiroyasu; et al.
International Journal of Environmental Analytical Chemistry, 86(9), p.663 - 675, 2006/08
Times Cited Count:14 Percentile:40.18(Chemistry, Analytical)The IAEA introduced the environmental sample analysis method, as a powerful tool to detect undeclared nuclear activities, into strengthened safeguards system. The principle of the method is that nuclear signatures can be evidenced if trace amount of nuclear materials in environmental samples taken from inside and outside of nuclear facilities are accurately analyzed. Currently, isotope ratios of uranium and plutonium in "swipe" samples are measured, which are collected in nuclear facilities. In future, the subject of environmental sample analysis will expand to soil, sediment, vegetation, water and airborne dust taken from outside of the nuclear facilities. If physical and chemical form of the nuclear materials is identified, we may estimate their origin, treatment process and migration behavior. This paper deals with the developed analytical techniques for the safeguards environmental samples, the current R&D on techniques related to estimation of the physical and chemical form, and possible analytical methodologies applicable to ultra-trace amounts of nuclear materials.
Lee, C. G.; Iguchi, Kazunari; Esaka, Fumitaka; Magara, Masaaki; Sakurai, Satoshi; Watanabe, Kazuo; Usuda, Shigekazu
Nuclear Instruments and Methods in Physics Research B, 245(2), p.440 - 444, 2006/04
Times Cited Count:8 Percentile:50.31(Instruments & Instrumentation)The etching rates of fission track detectors made of polycarbonate containing uranium particles were measured after thermal neutron irradiation with fluence of 8 10
10 n/cm
n/cm , in order to study the influence of uranium enrichment on the etching rate that was calculated from the weight loss by etching. There is a strong correlation between the etching rate of detector and the enrichment E of uranium particle: the former increases as the latter increases. Particularly, the etching rate per particle was proportional to E
, in order to study the influence of uranium enrichment on the etching rate that was calculated from the weight loss by etching. There is a strong correlation between the etching rate of detector and the enrichment E of uranium particle: the former increases as the latter increases. Particularly, the etching rate per particle was proportional to E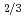 rather than E
rather than E , which is probably due to the overlapping of fission tracks. The etching behaviors of detector revealed that the existence of two different etching rate regions, a nonlinear region in the beginning of etching process and a subsequent constant region, which was explained as the opening of fission tracks and the broadening of opened tracks, respectively.
, which is probably due to the overlapping of fission tracks. The etching behaviors of detector revealed that the existence of two different etching rate regions, a nonlinear region in the beginning of etching process and a subsequent constant region, which was explained as the opening of fission tracks and the broadening of opened tracks, respectively.
Iguchi, Kazunari; Esaka, Konomi; Lee, C. G.; Inagawa, Jun; Esaka, Fumitaka; Onodera, Takashi; Fukuyama, Hiroyasu; Suzuki, Daisuke; Sakurai, Satoshi; Watanabe, Kazuo; et al.
Radiation Measurements, 40(2-6), p.363 - 366, 2005/11
Times Cited Count:11 Percentile:59.93(Nuclear Science & Technology)In particle analysis for safeguards environmental samples, the fission track technique is very important to detect sub-micrometer particles containing uranium. In the technique the authors developed, the particles were recovered onto the polycarbonate membrane filter. The filter was dissolved in solvent and dried to form a thin film of detector, in which the particles were confined. After thermal neutron irradiation and etching, the particles of interest in the detector were easily identified with fission tracks, and were picked up for isotope ratio analysis. It was found, however, that the particles in the vicinity of the detector surface may fall off during the etching process. Therefore, optimization of the etching condition is required. In this work, the effects of etching time and enrichment of uranium in particles were investigated. Preliminary results suggest that etching time should be shorter with the increase in the enrichment.
Lee, C. G.; Iguchi, Kazunari; Esaka, Konomi; Magara, Masaaki; Esaka, Fumitaka; Sakurai, Satoshi; Watanabe, Kazuo; Usuda, Shigekazu
Analytica Chimica Acta, 517(1-2), p.215 - 220, 2004/07
Times Cited Count:4 Percentile:13.8(Chemistry, Analytical)In this study, we discussed the feasibility of solid sample ETV-ICP-MS for the particle analysis of the environmental swipe samples in order to establish the fission track-ETV-ICP-MS method. Samples used were solid samples containing Tl, Pb and U in polycarbonate film, and their standard solutions. The analytical performance will be reported in terms of the optimization of temperature program, and sensitivity and precision in isotope ratio measurement.
Esaka, Konomi; Esaka, Fumitaka; Inagawa, Jun; Iguchi, Kazunari; Lee, C. G.; Sakurai, Satoshi; Watanabe, Kazuo; Usuda, Shigekazu
Japanese Journal of Applied Physics, Part 2, 43(7A), p.L915 - L916, 2004/07
Times Cited Count:41 Percentile:78.95(Physics, Applied)An effective method for isotope ratio analysis of individual particles containing uranium in safeguards swipe samples was developed by using a fission track (FT) technique combined with thermal ionization mass spectrometry (TIMS). The particles in the sample were directly recovered onto a polycarbonate membrane filter, which was set in a filtration system. After the particle recovery, the filter was dissolved to form a non-coated FT detector film containing the particles. Individual particles containing uranium were then identified by the observation of fission tracks created by thermal neutron irradiation. By combination of the FT technique with TIMS, isotope ratios of uranium for individual particles in the swipe sample prepared from a uranium standard reference material could be effectively determined.
Ikezoe, Yasumasa; ; Shimizu, Saburo
Radiation Physics and Chemistry, 21(3), p.341 - 344, 1983/00
no abstracts in English
 on fission tracks in polyvinylidene fluoride
on fission tracks in polyvinylidene fluoride; ; ;
J.Electron Microsc., 31(2), p.191 - 193, 1982/00
no abstracts in English
Ikezoe, Yasumasa; ; Shimizu, Saburo
Canadian Journal of Chemistry, 59(3), p.532 - 536, 1981/00
Times Cited Count:5 Percentile:42.27(Chemistry, Multidisciplinary)no abstracts in English
;
J.Electron Microsc., 30(4), p.292 - 297, 1981/00
no abstracts in English
 O
O ; Possible chemical form of iodine
; Possible chemical form of iodineNakashima, Mikio; Tachikawa, Enzo
Journal of Inorganic and Nuclear Chemistry, 43, p.1686 - 1687, 1981/00
Times Cited Count:1 Percentile:11.9(Chemistry, Inorganic & Nuclear)no abstracts in English
Ikezoe, Yasumasa; ; Shimizu, Saburo
Radiation Physics and Chemistry, 17, p.63 - 67, 1981/00
no abstracts in English
Nucl.Tracks, 13(1-2), p.33 - 44, 1979/00
no abstracts in English
;
Science, 199(4327), p.421 - 422, 1978/00
Times Cited Count:14no abstracts in English
Ikezoe, Yasumasa;
Journal of Nuclear Science and Technology, 13(9), p.503 - 507, 1976/09
Times Cited Count:11no abstracts in English
;
Journal of Applied Physics, 47(4), p.1355 - 1358, 1976/04
Times Cited Count:13no abstracts in English
; ; Izui, Kazuhiko;
Journal of Applied Physics, 46(3), p.1155 - 1158, 1975/03
Times Cited Count:6no abstracts in English
;
JAERI-M 5578, 33 Pages, 1974/03
no abstracts in English
Journal of Nuclear Science and Technology, 10(8), p.515 - 516, 1973/08
no abstracts in English